Validation Protocols: Setting up Approvals and Sign-offs - Tip193
OBJECTIVE or GOAL
Get Empowered: Method Validation Manager
Tip #193: Validation Protocols: Setting up Approvals and Sign-offs
The previous tip (Tip #192) continued the discussion on Validation Protocols and how to set up the System Precision test.
This tip continues the discussion on Validation Protocols and how to set up Approvals and Sign-offs.
You can configure each validation protocol method to enforce approval requirements, sign-off requirements, or both, during various stages of your method validation workflow. Approval and sign-off capabilities enable you to establish quality control checkpoints that your SOPs may require. For example, you might want a qualified staff member to review and approve all protocol methods before using them to create a validation study. You might also want all chromatographic results reviewed and signed-off before using them to create validation results within a study.
ENVIRONMENT
- Empower
PROCEDURE
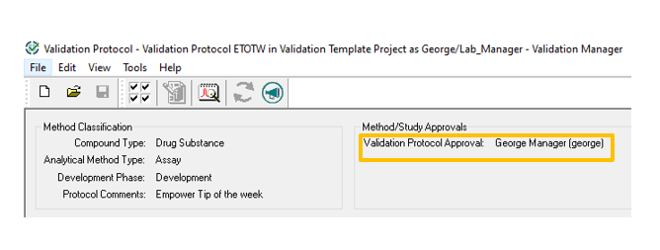
- Click the Approvals/Sign Offs tab in the Validation Protocol. Note that the Validation Protocol Approval field is empty.
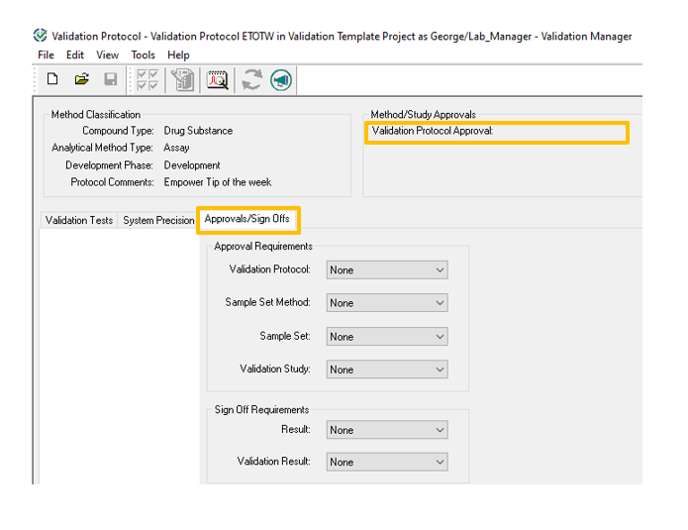
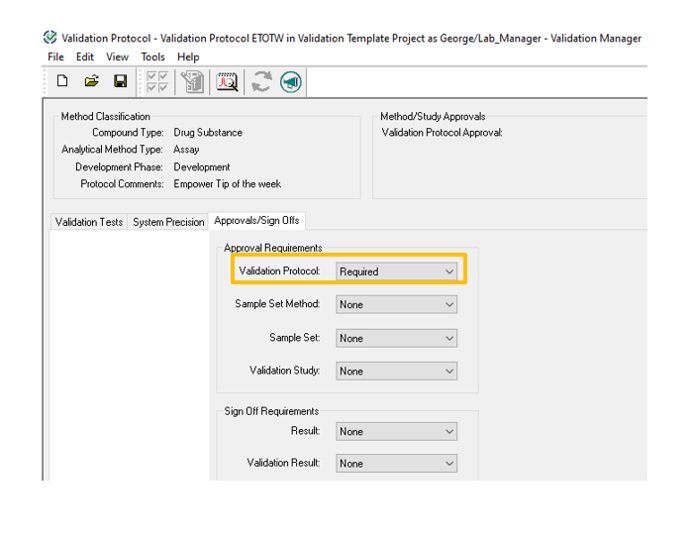
- From the Validation Protocol drop-down list, select Required.
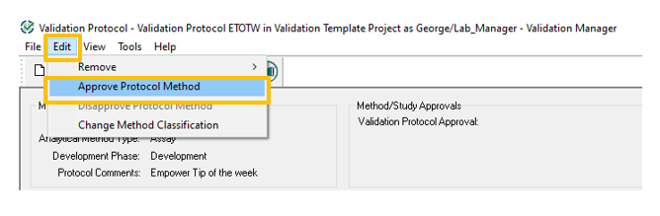
- To approve the protocol, click the Edit pull-down menu and select Approve Protocol Method.
- Enter your username and password, and then click OK.
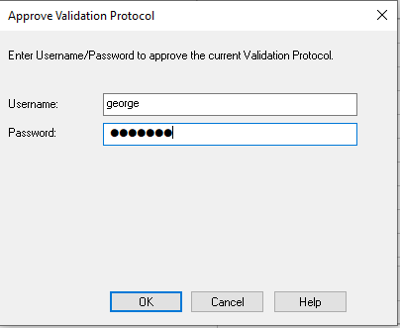
- The Validation Protocol Approval field is populated. You can now create a new validation study using this protocol.
ADDITIONAL INFORMATION
Use the Pro interface when working with the Method Validation Manager option.

